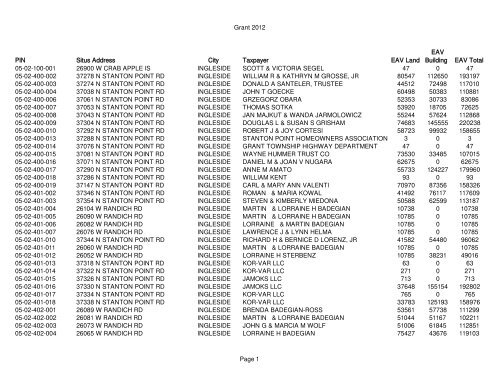
- View & download of more than 1495 Bunn PDF user manuals, service manuals, operating guides. Coffee maker user manuals, operating guides & specifications.
- This section is dedicated to Oxygen Equipment manuals & user guides which are included in the main list of categories. The page provides a catalogue of brands and devices, each offering to view or download an updated manual. To see the entire list of Oxygen Equipment items designed by a particular manufacturer click on ‘More’ button.
- This user manual contains important information and safety precautions for the John Bunn Neb-u-Lite EV Nebulizer Compressor. Before using your Nebulizer Compressor, please read and understand this entire user manual. Take special note of all safety precautions marked DANGER and WARNING. Save this user manual for future reference. Intended Use.
- User ManUal Model numbers JB0160-010/JB0160-010-220 and JB0160-015/JB0160-015-220 with O 2. The John Bunn O2 Premier oxygen concentrator is intend-ed for use as an oxygen supplement device in a home or care facility. Use the concentrator only as prescribed by a physician. This user manual serves as a reference for your.
View online Operator's manual for John Bunn JB02017 Medical Equipment or simply click Download button to examine the John Bunn JB02017 guidelines offline on your desktop or laptop computer.
OXYCHECK
FINGERTIP
PULSE OXIMETER
MODEL JB02017
OPERATOR MANUAL
GENERAL DESCRIPTION
Bunn Parts Manual
The John Bunn® JB02017 OxyCheck Fingertip Pulse Oximeter provides a simple way to spot-check users by combining the sensor and monitor into one integrated, compact, easy to use device. The oximeter measures pulse oxygen saturation (SpO2 ) value, pulse rate value, and pulse strength. When a finger is inserted into the sensor’s rubber cushion, the SpO2 value automatically displays. The pulse bar graph displays the user’s pulse beat, and the bar graph’s height shows pulse strength. The oximeter, which is powered by two AAA batteries, features a lowbattery indicator and powers off automatically in eight seconds when not in use.
Product Accessories (Included)
1.One lanyard
2.Two AAA batteries
3.One protective cover
4.One operator manual
Principle of Measurement
Two beams of different wavelength (660 nm glow and 940 nm near infrared light) are focused onto a human nail tip through a clamping finger-type sensor. A measured signal obtained by a photosensitive element, through processes of electronic circuits and microprocessor, will be shown on the oximeter’s display.
Principle of Operation Diagram
See illustration and descriptions below.
1 | Red and Infrared Emission Tube |
2 | Red and Infrared Receipt Tube |
INTENDED USE
The intended use of the OxyCheck Fingertip Pulse Oximeter is the measurement and display of the functional oxygen saturation of arterial hemoglobin (SpO2) and pulse rate (PR) of adults and pediatric users in hospital, ambulatory, home, and EMS (Emergency Medical Service) environments. The Pulse Oximeter is intended for spot-checking these levels.
Contraindications ( WARNINGS):
•If you do not understand any part of these instructions, contact a healthcare professional for direction in the use of this product.
•This device is not intended for continuous monitoring.
•Do not use this device in an explosive atmosphere.
•Do not use this device in an MRI or CT environment.
INACCURATE MEASUREMENTS MAY BE CAUSED BY THE FOLLOWING:
•Autoclaving, ethylene oxide sterilizing, or immersing the device in liquid
•Significant levels of dysfunctional hemoglobins (such as carbonxyhemoglobin or methemoglobin)
•Intravascular dyes such as indocyanine green or methylene blue
•High ambient light — shield the sensor area (with a surgical towel, or direct sunlight, for example) if necessary
•Excessive user movement
•High-frequency electrosurgical interference
•Venous pulsations
•Placement of a sensor on an extremity with a blood pressure cuff, arterial catheter, or intravascular line
•User hypotension, severe vasoconstriction, severe anemia, or hypothermia
•User cardiac arrest or shock
•Improper finger placement, e.g. fingernail not facing upward
•Fingernail polish or false fingernails. | 1 |
SAFETY — PRECAUTIONS FOR USE
WARNING: Indicates a potential hazard situation or unsafe practice that, if not avoided, could result in death or serious injury. WARNING statements follow:
Before use, carefully read the manual.
The pulse oximeter has no alarms. Do not use the pulse oximeter in situations where alarms are required. It is not for continuous monitoring.
The pulse oximeter is intended only as an adjunct in user assessment. It must be used in conjunction with other methods of assessing clinical signs and symptoms.
CAUTION: Indicates a potential hazard or unsafe practice that, if not avoided, could result in moderate or minor personal injury. CAUTION statements follow:
Check the pulse oximeter sensor application site frequently to determine the positioning of the sensor and circulation and skin sensitivity of the user.
Do not stretch the adhesive tape while applying the pulse oximeter sensor. This may cause inaccurate readings or skin blisters.

Prolonged use or the user’s condition may require changing the sensor site periodically. Change sensor site and check skin integrity, circulatory status, and correct alignment at least every 4 hours.
s NOTICE: Indicates a potential hazard or unsafe practice that, if not avoided, could result in product/property damage.
SETUP
Battery Installation
1.Open battery compartment cover.
2.Install two AAA batteries in battery compartment, ensuring that polarities are correct.
3.Close battery compartment cover: Push cover horizontally along the arrow as shown at right.
s NOTICE:
•Ensure battery polarities are correct, or the device could be damaged.
Lanyard Installation
1.Thread the thinner end of the lanyard through the oximeter loop.
2.Thread the thicker end of the lanyard through the threaded end, then pull it tightly.
OPERATION INSTRUCTIONS
1.Use isopropyl alcohol to clean the test finger and the rubber inside the oximeter that touches the finger.
2.Place clamp over fingernail as shown at right; insert finger, fingernail up as shown.
3.Press button on front panel once. User’s finger and body must remain still during measurement.
4.See display: the SpO2 value automatically displays, the pulse bar graph displays the pulse rate, and the bar graph’s height shows the pulse strength.
MAINTENANCE AND STORAGE s NOTICE:
•This device contains no serviceable parts. Do not disassemble.
•Remove the batteries if the oximeter will not be used for a long period of time.
•Do not autoclave, sterilize with ethylene oxide, or immerse the device in liquid.
•See SPECIFICATIONS/Environmental Requirements for operation and storage requirements. A wet ambience could damage this product and shorten its lifetime.
•Recycle or dispose of this device and its used batteries in observance of local regulations.
Info: Use isopropyl alcohol to clean the rubber (inside the oximeter, that touches the finger) and the test finger before and after each test. The rubber inside the oximeter is medical rubber, which has no toxins, and is not harmful to the skin.
•Replace the batteries when low battery indicator illuminates.
SPECIFICATIONS
Display Type | LED (Light Emitting Diode) | ||||||
SpO2 | Measurement | 70-99% | |||||
range | |||||||
Accuracy | 80%-99%: ±2% | ||||||
70%-79%: ±3% | |||||||
≤69% : no definition | |||||||
Pulse Rate | Measurement | 30-235 BPM | |||||
range | |||||||
Accuracy | 30~99 BPM: ±2 BPM | ||||||
100~235 BPM: ±2% | |||||||
Pulse Intensity | Bargraph Indicator | ||||||
Power Requirement | Two AAA alkaline Batteries | ||||||
Power Consumption | <40 mA | ||||||
Low Power Indicator | |||||||
Battery Life | ~ 30 hours of continuous operation | ||||||
Dimension | 2.20″ ~ 2.44″ x 1.26″ ~ 1.50″ x 1.34″ ~ 1.50″ | ||||||
(L x W x H) | (56 mm ~ 62 mm x 32 mm ~ 38 mm x 34 mm ~ 38 mm) | ||||||
Weight | 1.59 oz ~ 2.12 oz (0.10 lb ~ 0.13 lb) (45 g ~ 60 g) | ||||||
including two AAA batteries | |||||||
Environmental | Temperature | Operation | 41°F ~ 104°F | ||||
Requirements | (5°C ~ 40°C) | ||||||
Storage | 68°F ~ 131°F | ||||||
(20°C ~ 55°C) | |||||||
Humidity | Operation | ≤80% RH | |||||
(non-condensing) | |||||||
Storage | ≤93% RH | ||||||
Interference | Device works normally when mixed noise produced | ||||||
Resistance Capacity | by BIO-TEK INDEX Pulse Oximeter tester | ||||||
againstAmbient Light | |||||||
DECLARATION
This product’s EMC complies with IEC60601-1-2 standard.
The materials with which the user can come into contact have no toxicity, no action on tissues, and comply with ISO10993-1, ISO10993-5 and ISO10993-10.
GUIDANCE AND MANUFACTURER’S DECLARATION: ELECTROMAGNETIC EMISSIONS FOR ALL EQUIPMENT AND SYSTEMS
Guidance and manufacturer’s declaration – electromagnetic emission
The Pulse Oximeter is intended for use in the electromagnetic environment specified below. The customer or the user of the Pulse Oximeter should assure that it is used in such an environment.
EmissionTest | Compliance | Electromagnetic environment – guidance | ||||
RF emission | Group 1 | The Pulse Oximeter uses RF energy only for its | ||||
CISPR 11 | internal function. Therefore, its RF emissions | |||||
are very low and are not likely to cause any | ||||||
interference in nearby electronic equipment. | ||||||
RF emission | Class B | The Pulse Oximeter is suitable for use | ||||
CISPR 11 | in all establishments, including domestic | |||||
establishments and those directly connected to | ||||||
the public low-voltage power supply network that | ||||||
supplies buildings used for domestic purposes. | ||||||
TROUBLESHOOTING | ||||||
Problem | Possible reason | Solution | ||||
SpO | 2 | or PR | 1. User's finger is incorrectly | 1. Reinsert user's finger | ||
inserted | 2. Attempt several times to | |||||
cannot be | ||||||
displayed | 2. User’s Oxyhemoglobin | obtain a reading; If sure that | ||||
normally | value is too low to be | no problem exists, obtain | ||||
measured | further clinical examination | |||||
SpO | 2 | or PR | 1. Finger may not be | 1. Reinsert finger | ||
inserted deeply enough | 2. Ask user to remain still | |||||
display is | ||||||
unstable | 2. Finger trembling or user | |||||
moving | ||||||
Troubleshooting continued | ||||||||||
Problem | Possible reason | Solution | ||||||||
Oximeter | 1. | Battery power may be | 1. | Replace batteries | ||||||
cannot be | inadequate or batteries | 2. | Reinstall batteries | |||||||
powered on | may not be installed | 3. | Contact GF distributor | |||||||
2. | Batteries may be installed | |||||||||
incorrectly | ||||||||||
3. | Oximeter may be damaged | |||||||||
Indicator | 1. | Device automatically | 1. | Normal | ||||||
lamps are | powers off when no signal | 2. | Replace the batteries | |||||||
suddenly | is detected for longer than | |||||||||
off | 8 seconds | |||||||||
2. | Batteries too weak to | |||||||||
power device | ||||||||||
“Error3” or | 1. | Low power | 1. | Replace batteries | ||||||
“Error4” | 2. | Receiving tube and/or | 2. | Contact GF distributor | ||||||
displays | connector may be | 3. | Contact GF distributor | |||||||
shielded or damaged | 4. | Contact GF distributor | ||||||||
3. | Mechanical misplace for | |||||||||
receive-emission tube | ||||||||||
4. Amp circuit malfunction | ||||||||||
“Error7” | 1. | Low power | 1. | Replace batteries | ||||||
displays | 2. | Emission diode damaged | 2. | Contact GF distributor | ||||||
3. | Current control circuit | 3. | Contact GF distributor | |||||||
malfunction | ||||||||||
SYMBOL DEFINITIONS | ||||||||||
Symbol | Definition | Symbol | Definition | |||||||
Type BF applied part | Low power indicator | |||||||||
Attention, consult | No SpO2 Alarm | |||||||||
accompanying documents | ||||||||||
SpO2 | Oxygen saturation | | Power switch | |||||||
Heart rate (BPM) | SN | Serial Number | ||||||||
BPM | ||||||||||
Info: The illustration used in this manual may differ slightly from the appearance of the actual product.
LIMITED WARRANTY
GF Health Products, Inc. warrants the John Bunn® JB0217 OxyCheck Fingertip Pulse Oximeter to be free from defects in workmanship and materials for one year.
The warranty does not apply to damage resulting from failure to follow the operating instructions, accidents, abuse, alterations or disassembly by unauthorized individuals.
If a product is deemed to be under warranty, GF Health Products, Inc. shall provide, at its option, (1) replacement of any defective part or product or (2) a credit of the original selling price made to GF Health Products, Inc.’s initial customer. The warranty does not include any labor charges incurred in replacement part(s) installation or any associated freight or shipping charges to GF Health Products, Inc.
The warranties contained herein contain all the representations and warranties with respect to the subject matter of this document, and supersede all prior negotiations, agreements and understandings with respect thereto. The recipient of this document hereby acknowledges and represents that it has not relied on any representation, assertion, guarantee, warranty, collateral contract or other assurance, except those set out in this document.
www.grahamfield.com
Bunn Manuals Pdfs
Graham-Field and John Bunn are registered trademarks of GF Health Products, Inc.
Packaging, warranties, products, and specifications are subject to change without notice. GF Health Products, Inc. is not responsible for typographical errors.
© 2010 GF Health Products, Inc., Atlanta GA 30360, 770-368-4700 | JB02017-INS-LAB-RevA10 |
To continue the download you need to go through the captcha:
